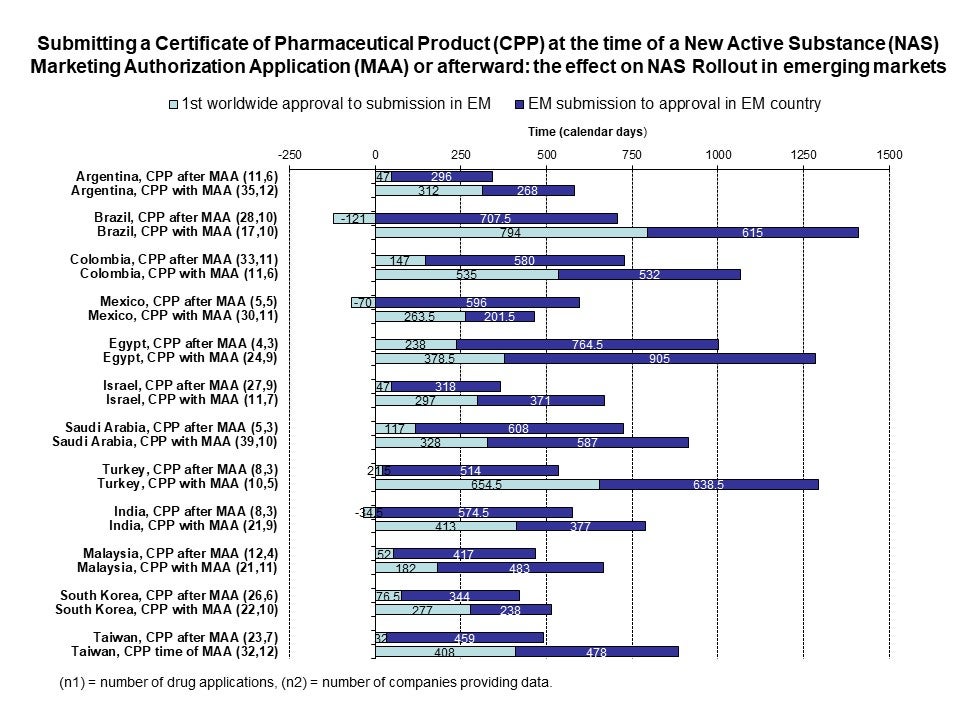
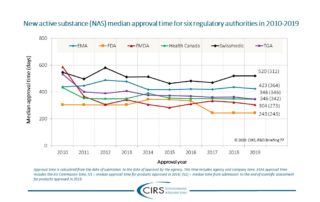
Liberti et al 2020 – Evaluation of the Caribbean Regulatory System centralised assessment process
Background: The Caribbean Regulatory System is a centralized medicine assessment procedure established to serve the needs of the Member States of the CARICOM region. In order to better understand [...]