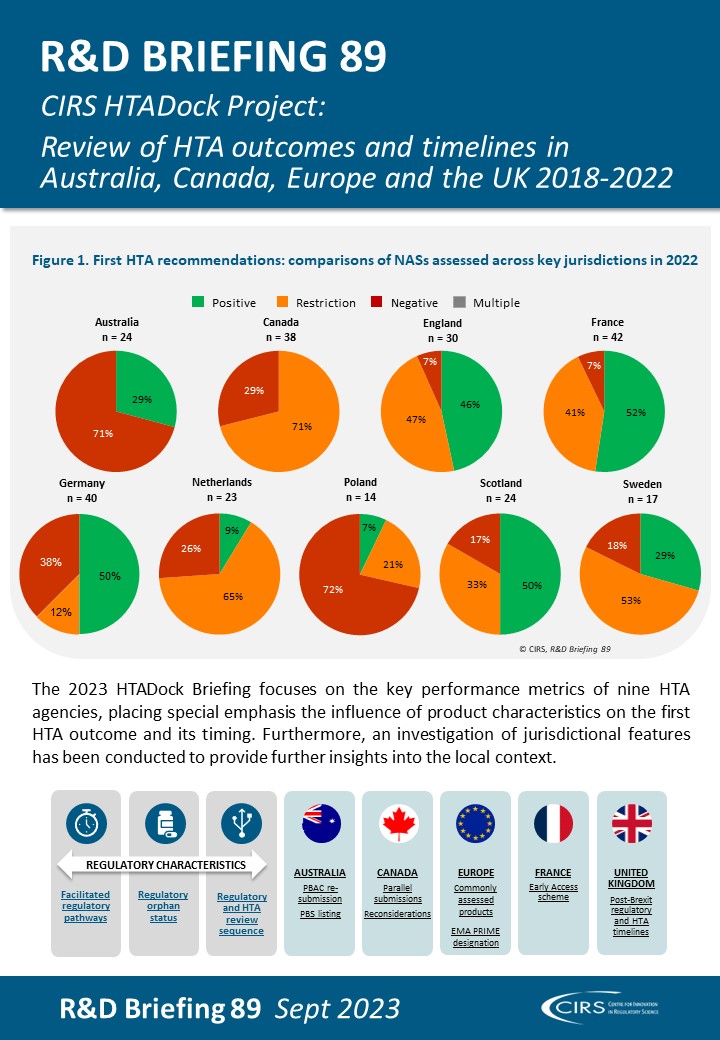
Rare Disease Product Approvals: The Changing Regulatory And HTA Landscape Between 2018-2022
Background Globally, 7,000 rare diseases affecting 300 million people pose development challenges with small patient populations. Developing medicines for rare diseases requires innovation. Despite regulatory incentives, challenges for HTA [...]